| |
 |
| WHAT TYPE OF FACE MASK IS THE MOST EFFECTIVE AGAINST THE CORONAVIRUS |
 |
| |
|
|
|

|
|
The covid-19 has disrupted every facet of life on the planet, from finance, to health and even our greeting and lifestyles. To combat the spread of Coronavirus, there has been a widespread campaign by governments and health organizations for the use of face masks alongside other preventive measures like social distancing and washing of hands.
These face masks come in different types – cloth masks, surgical masks, N95 masks – and this article will examine each type of face mask to help you decide on which is the best and most suitable to protect you during the pandemic.
N95 masks shall be reserved to the healthcare workers
N95 masks, also called N95 respirators, are NIOSH approved face masks that filter out 95% of all droplets and aerosols of size between 0.1 and 0.3 microns in width – including droplets that carry the novel coronavirus. They are tight fitting and provide more protection than other types of masks. Health professionals are usually fit tested in the production of these masks to ensure adequate sealing. Even though research is being carried out to make N95 masks reusable, they are still single-use face masks.
Due to the limited supply of N95 masks in some countries, the general public has been urged to reserve its usage for health workers and first line responders who are more at risk of coronavirus.
N95 masks generally increase the effort needed for breathing which might lead to suffocation in younger children (below 3 years) with smaller lung capacities. Also, N95 masks are generally designed to fit adults and might not fit children less than 12 years of age.
People with medical conditions such as chronic lung and heart diseases should consult a doctor before wearing N95 face masks. This is because they might already have features of a reduced lung volume and wearing the face masks can make breathing more difficult and uncomfortable.
Surgical masks are the best types of masks for the general public
Surgical masks or medical-grade face masks are disposable, light blue, loose fitting masks.
They usually have strings that are tied behind the head, or elastic bands that fit over both ears. Surgical masks usually consist of three layers and they protect the nose and mouth of the wearer from droplet transmission.
Unlike the N95, they cannot protect against aerosols, and since they are loose fitting, they do not provide complete protection from potential contaminants. But they still serve as a potent physical barrier between the mouth and nose of the wearer and sprays, droplets or splashes.
“When used correctly, surgical masks help in preventing the spread of covid-19. When worn, the blue part should face outside while the white part should face the mouth and nose. Combining them with filters or cloth masks can also increase their effectiveness”, reminds Michelle Chiu from The Kare Lab, a manufacturer in medical supplies.
Surgical masks are designated for single-use and should be properly and safely discarded after use.
Handmade and clothes masks
Handmade and cloth masks became a necessary invention during the pandemic as the world was facing a face mask shortage due to the high demand.
They come in different types, and sizes. An effective cloth mask should have at least three layers of fabric because higher protection is provided by more layers of fabric in the mask.
Against small particles, cloth masks are not as effective as respirators or surgical masks, but they are able to block slightly larger particles and also help in preventing the spread of coronavirus.
The most significant advantage of cloth masks is that they are reusable after washing. However, it is important that they are washed properly as some studies suggest that proper laundering helps them perform better against smaller particles.
They are also very easy to make or purchase and they can be combined with surgical masks for better protection. In using cloth masks, the nose, mouth and chin should be well covered and the wearer should avoid touching them.
Other masks
Apart from the masks mentioned above, there are several other types. Some include:
• KN95 masks are similar to the N95 but are made in China. They also filter 95% of all droplets and aerosols. Only KN95 masks that meet NIOSH requirements should be purchased.
• FFP2 masks are European products that provide at least 94% filtration. They are similar to the N95 masks.
• Masks with valves have valves that make breathing more comfortable but they are not as effective in limiting the spread of viruses since they allow expelled hair through the holes, which can transmit droplets to others.
• Plastic face shields and googles are effective only as additional protection to face masks. They do not offer enough protection against coronavirus because there is still a large space between the shield and the mouth and nose through which droplets can enter but they represent a perfect item in completion with the mask.
|
|
|
| |
|
|
| |
|
|
|
|
|
|
|
|
|
 |
 |
| Journey Of Alcohol In Your Body |
 |
| |
|
|
|
|
|
|
Introduction
How many of us have ever had one too many shots at a party, waking up in bed with a pounding in your head like there’s a sledgehammer going to town in your skull? That is just one of the many symptoms of alcohol use disorder and if you are here, you probably are looking for a solution to a night of overindulgence. The bad news is that it doesn’t exist. However, you can learn something here that will hopefully make your next drinking session a lot less painful and regretful.
How Long Does Alcohol Stay in Your Body?
So here’s the key question, what is alcohol? The average American drinks 2.3 gallons of alcohol a year, but never stopped to consider how this depressant makes its way through the body (and brain). Like everything else that is eaten or drunk, alcohol is broken down by the body for energy through metabolism. An interesting fact is that once it permeates the bloodstream, the body metabolizes it at a constant speed of 20 mg/dL every hour. This means that it would take exactly one hour to break down 200 milligrams of alcohol in a liter of blood. Going by the same logic, it would take three hours to metabolize 600 mg of alcohol in a liter of blood.
If you are asking why everybody is not equally wasted at the end of a party if we drank the same number of beer pints? The answer lies in the blood alcohol concentration (BAC) of your blood. The blood alcohol concentration is simply the ratio of alcohol to water in your blood. A higher ratio indicates a higher level of intoxication, which increases the side effects of alcohol on your system. Therefore, it is a given to drink plenty of water in order to reduce BAC levels Even though two people can both have exactly 20mg/DL for their blood alcohol level, they can have the different BAC levels, which will result in differing levels of drunkness.
The most common factors that influence BAC are weight, binge drinking, and drinking on an empty stomach. That being said, age, liver disease, and the medication you are on also affect BAC levels. Do take note that different drinks take differing amounts of time to metabolize completely. A shot of hard liquor takes an hour, a pint of beer takes double that and a glass of wine will require around three hours to metabolize.
How it is Metabolized
For all the talk of alcohol in blood, we have yet to cover on how it enters the bloodstream from your mouth. Roughly 20 percent of your drink goes in your blood vessels and straight to the brain. The other 80 percent actually follow the path of food, entering the esophagus ending its journey in the small intestine. It then enters the bloodstream and is eventually removed by the liver. This is why people with liver issues tend to have higher levels of BAC in general as their systems are slower in removing the alcohol from their system.
Alcohol Detection Tests
To measure the levels of BAC, there are numerous tests that can be conducted. The most common of which is the breath test through a breathalyzer. This device collects the breath of the person being tested and measures the BAC levels instantly. A person with BAC levels greater than 0.02 should not operate heavy machinery or drive in order to prevent safety accidents. Alcohol can be detected through the breath test within 24 hours. For a test that can detect alcohol within the system up to 80 hours after drinking, the urine test is a great alternative if you are looking at more detailed testing methods. However, simpler methods can also detect alcohol up to 48 hours after consumption. This is mostly attributed to the fact that traces of alcohol can be detected for up to 90 days in a person’s hair, and this fact has been used in forensic investigations.
Alcohol and Breastfeeding
Everybody knows that alcohol is to be avoided during pregnancy, but did you know that it affects breastfeeding and the child being breastfed? Alcohol can be transferred to the baby through the umbilical cord and can cause physical and intellectual disabilities. Alcohol in breast milk can cause the same effects and development issues when the baby is breastfed by his mum who had recently consumed alcohol. For breastfeeding mums out there, it is better to abstain from consuming alcohol entirely. If alcohol is to be consumed by the breastfeeding mum, it would be better to pump out and store milk in advance as well as to feed the baby before drinking. Most importantly, after drinking a shot of liquor or a small glass of wine, it is best to stagger a period of three hours before breastfeeding the baby.
Alcohol Poisoning
Finally, we have come to the last but most important part of the article, which is regarding alcohol poisoning. It can be fatal and should be considered a medical emergency. This usually happens when there is an excessive amount of alcohol consumed and when it cannot be metabolized sufficiently quickly. A common cause of alcohol poisoning is binge drinking which causes the victim to continuously imbibe alcohol without understanding the full consequences of their actions.
If the victim passes out, vomits, has a lower blood temperature, and is suffering from slower breathing, there is a good chance that the person is suffering from alcohol poisoning. In the event of such an emergency, an ambulance should be called for and the victim should not be left unattended. While waiting for the ambulance, the victim should be lying on their side to avoid choking on their own vomit.
Conclusion
This article has explored the journey alcohol takes in our body, as well as how long it stays and how it affects us when over-consumed. Drinking can remain a fun and safe social activity when done responsibly and this article hopes to impart some knowledge regarding that.
|
|
|
| |
|
|
| |
|
|
|
|
|
|
|
|
|
 |
 |
| What to Do When Your Medical Care Goes Wrong |
 |
| |
|
|
|
|
|
|
Although medical care facilities and services have a vast number of quality checks to ensure that each patient receives the best treatment possible for their condition, it is still possible that your treatment that can wrong. This can be due to a number of factors, including human error and negligence, and can have a large impact on your condition’s ability to improve. If you have been given treatment that has worsened your condition, there are a number of steps that you can take to get the justice that you deserve.
1. Complain to the Hospital or Care Provider
The first step that you should take for both minor and more serious complaints is to complain directly to the hospital where you received treatment. This is especially important if treatment is ongoing as this will allow your situation to be rectified as soon as possible. In a public healthcare facility, they will have their own independent written and verbal complaints procedures which can help you to get the resolution that you need. Your complaint is legally obliged to be investigated, and you will get a written response in reply to this complaint.
2. Get a Lawyer
If your case is not resolved, you should consider getting a lawyer, who can help you to receive the compensation that you need and guide you through the court process if the situation escalates. IBB Claims can help you to rectify a wide variety of medical negligence claims, such as paramedic negligence, as their trained team of advisors can help you to collect evidence, discuss the strength of your advice, and give you advice as to the next steps that you need to take in order to get the end result that you desire.
3. Speak to Your Country’s Health Ombudsman
You can also discuss your case with a health or public ombudsman, if your country has one, which is a regulatory body that is created to investigate the public’s complaints with an organization. They will lead an investigation into your case if your complaint does not give you the answer that you were looking for. They will be able to compare the care that you received with the hospital’s policy, check through medical records and documents, and ascertain blame if needed.
4. Speak to Your Insurer
It is also important that you speak to your insurance provider if you gained hospital or paramedic care from a private health company. This is especially important if your complaint is in reference to your health providence policy or the execution of one of their policies, such as if you don’t have the coverage that you expected. They will also be able to give you unbiased advice as to the next steps to take and who you should complain to.
5. Get a Second Opinion
However, you should never let the complaints procedure affect your medical health, and you should seek a second medical opinion if you are unhappy with the diagnosis or care that you received elsewhere. Not only this, but a medical examination can provide key evidence for any court cases that occur.
|
|
|
| |
|
|
| |
|
|
|
|
|
|
|
|
|
 |
 |
| All You Need To Know About Silent Heart Attacks |
 |
| |
|
|
|
|
|
|
Nowadays, silent heart attacks have become extremely common, and they can happen without the victim realising it.
According to Dr. Leslie Tay, an interventional cardiologist at Mount Elizabeth Hospital, silent attacks occur without visible symptoms, and there are some little signs you should watch out for.
What is a silent heart attack?
In a heart attack, one of the heart arteries become blocked to the extent where blood flow is diminished. As a result of which, the heart muscle becomes deprived of nutrients and oxygen, gets destructed and eventually dies.
The most common symptoms of heart attack are severe crushing chest pain or heaviness. The pain suffered in a heart attack is described as an elephant sitting on your chest, along with cold sweats and nausea.
On the other hand, in a silent heart attack, the patient does not experience these full-blown symptoms. In fact, they may feel nothing at all. All they experience are a few less subtle symptoms which are usually mistaken for a less critical condition.
Causes of a silent heart attack
A silent heart attack happens for the same reason as that of a normal heart attack. Over the years, the heart arteries tend to narrow from deposits of plaque and cholesterol. These deposits can anytime bleed, rupture and block off the artery. This leads to heart muscle damage.
Are silent heart attacks common?
As per research, almost half of all heart attacks are silent. Apparently, most people are not aware of their critical condition. Since they don’t seek treatment, they have a higher risk of dying.
Hence, one needs to be extremely cautious if they have any illness which causes heart attack, like diabetes. The nerve cells of diabetics are so damaged that do not experience pain the same way as someone with no diabetes. These people are more prone to having a silent heart attack; this is because of poor circulation and high sugar in their blood.
How to know if you can have a silent heart attack?
Some of the subtle signs of silent attack include discomfort in the chest back, arms or jaw. Apart from that, they may experience fainting spells
or dizziness. Also, running up a flight of stairs could wear them out. They might also have an upset stomach or heartburn sensation.
I have had some patients who have had a silent heart attack. One patient claimed that he had a severe gastric and heartburn sensation lasting over two days, accompanied by an overwhelming sense of fatigue. By the time the person made it to the hospital, his heart muscle was so damaged that there was an apparent heart failure. To recover the heart muscle, we had to open up the blocked artery.
You know your body best, and if at some point you feel that something isn’t right, then you should seek treatment as soon as possible.
What if I don't seek medical help immediately?
If you feel that something is not right in your body, you should get it checked out immediately. A heart attack is a severe condition which can potentially lead to death.
How can an interventional cardiologist help?
Apart from treating patients with heart diseases, cardiologists also manage risk factors like cholesterol and hypertension. In the case of a heart attack, an interventional cardiologist performs a procedure to open up the heart arteries. This is done to stop the heart attack and restore blood flow.
How can I prevent a silent heart attack?
Here are three of the most effective approaches:
1. Identify any existing artery blockages by going for a health screening get treated if necessary.
2. If there are any risk factors like obesity, smoking, diabetes, high cholesterol and high blood pressure, then keep them under control with the help of your doctor. Follow the medication your doctor prescribes medication and quit smoking.
3. Eat healthily and exercise regularly. Exercising for just 20 minutes every day can significantly reduce your risk of death.
In conclusion, it’s crucial to go for health screening. This will help you not only identify your risk factors, but also keep them under control. Adopting a healthy lifestyle with healthy eating and exercise will minimise your risk of developing a heart attack.
The inspiration for this article has been taken from Health Plus Section, a source of credible health information by Mount Elizabeth Hospitals, Singapore.
|
|
|
| |
|
|
| |
|
|
|
|
|
|
|
|
|
 |
 |
| Telemedicine Gaining in Popularity in Asia according to Report |
 |
| |
|
|
|
|
|
|
Telemedicine has been expanding in the rural parts of the United States rapidly due to the difficulty of finding medical professionals with an online HIM degree willing to live there and the cost savings that it brings when transportation isn’t required for homebound patients to be checked. Telemedicine is also gaining in popularity in Asia according to reports. Let’s look at the reasons why.
Rural Service in China
China has been rolling out telemedicine to bring skilled medical expertise to rural areas without having to transport them professionals across the wide, often mountainous country. China’s National health and Family Planning Commission started setting up a telemedicine network in 2013 to connect local health workers to remote experts with a bachelors in health information management. When the network is fully deployed and functional, it will be the largest telemedicine system on the planet.
China is also using the telemedicine network to send test results from EKGs and ultrasound probes generated by medical assistants to trained experts, allowing patients to receive expert opinions without having to travel to hospitals in major cities. Singapore’s mClinica is working on equipment to support this endeavor to bring professional care to rural villages and poor neighborhoods served by people with relatively little medical training.
Japan’s Efforts to Support Aging in Place
Japan is developing telemedicine networks to monitor many elderly who are homebound or have limited ability to travel at low cost. They are looking at ways to reduce medical costs in a nation with a shrinking population due to 30 years of below replacement birth rates. Eliminating the need to send nurses to visit the growing aging population saves everyone time and saves Japan’s universal healthcare system money while its economy remains in the doldrums. Singapore and South Korea are experiencing a similar “gray wave” and are looking at using telemedicine to reduce the cost of care to monitor the elderly and minimize the need for hospital stays.
The benefits for patients are obvious as well, such as stroke rehabilitation patients in Singapore using iPads for rehab instead of trying to arrange travel to a clinic. The government doesn’t need as many therapists, patients don’t have to leave their homes, and patients’ caregivers aren’t burdened by the effort of getting them to and from therapy. The cost of this service is a tenth that of conventional rehabilitation. Tele-rehab doesn’t work for everyone, but saving time and money for some patients leaves more resources for everyone without sacrificing quality of care.
Regional Awareness of Cost Savings
In Asia, the telemedicine, remote patient monitoring, electronic health records and mobile health networks are expected to approach two billion dollars by 2020. The annual growth rate is over 12%, driven by nations investing in lower cost, less resource intensive care for the under-served and the greater efficiency of limited numbers of medical staff to care for the elderly.
They can now afford to be proactive instead of struggling to meet basic medical needs for the population. And better connectivity and longer life expectancies mean they have both the reasons and resources to adopt telemedicine. The value is so well defined that telemedicine is part of the basic Smart City plan and Smart City infrastructure, which many Asian cities are implementing so they can modernize as quickly and effectively as possible.
|
|
|
| |
|
|
| |
|
|
|
|
|
|
|
|
|
 |
 |
| Modern Tech Is Empowering People with Disabilities |
 |
| |
|
|
|
|
|
Advancements over the past two decades have helped technology adapt to the needs of people with disabilities. According to a report, there are over 650 million disabled individuals in Asia and the Pacific. These individuals are benefiting from the discovery that programmable technology has facile capacity to accommodate human needs for assistance.
Currently, companies are using the application of physics and science to develop technology that helps improve the quality of life for individuals suffering from various disabilities like speech impairment, motion impairment, etc. As a result, assistive technology has become an umbrella term that includes everything from the tablets for the visually impaired to touch-free smartphones.
Overall, the use of modern tech in households with disabled people is on the rise. The following is a list of the latest technological equipment that is providing independence and freedom that came with restrictions in the past.
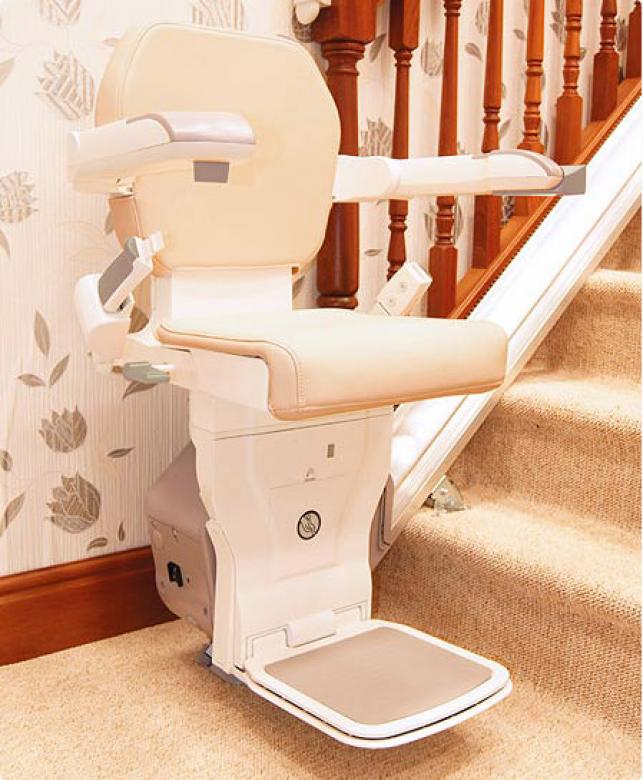
1. Stair Climber
Websites such as https://www.easyclimber.com/stair-lifts/ and other similar companies have introduced stairlift technology. It’s a chairlift device that allows people with physical disabilities to be seated as it goes up and down the flight of steps. Over time, advancements in stair lift technology have enabled the creation of sturdy designs that support attachment to the stair treads, allowing for quicker installation and no damage to surrounding walls. Most modern stairlifts can be completely custom made to allow for better fitting.
2. Personal Navigation Device
Wheelchair users face a range of issues, and loss of sight is one of them. Personal navigation devices like The Kapten Plus can make navigating around the sidewalks safe for such people. Modern navigation devices attach to the individual or the wheelchair. Then it relies on a GPS locator to provide directions and details about a particular location to users. This innovative tech provides wheelchair users with a peace of mind that they won’t get lost. It can also be used to find a safe spot nearby where wheelchair users can wait in and call someone for assistance.
3. Parkinson-Friendly Handle
Self-stabilizing handles like Liftware are allowing Parkinson patients to eat normally by attaching an eating utensil to the handle. It is quite useful for patients who are suffering from motion disorders that result in hand tremors. The self-stabilizing technology calms the movement which reduces the spilling of food content from the attached utensil before it goes into the patient’s mouth. The particular offering – liftware – features a stabilizing handle, three utensils (fork, spoon and soup spoon), and a charger. The charge lasts over several meals and users can wipe down the handle while the fork and spoon can be cleaned like normal utensils.
4. Wearable Finger-Worn Device
Wearable finger-worn devices like the FingerReader are being researched to enable the vision-impaired to convert any text into audio in dim lighting conditions. The technology makes use of haptic motors that vibrate when the user’s finger deviates from the text. A musical tone also goes off as an alert if the finger doesn’t track properly. There’s also a software and a tiny camera to process videos in real-time as the user sweeps his or her finger from left to right. Such technology may also be useful for individuals with dyslexia or learning disabilities.
The future of assistive technology looks bright as researchers and engineers around the globe continue designing and creating new devices based on different needs of the disabled.
Written Jane Brown
|
|
|
| |
|
|
| |
|
|
|
|
|
|
|
|
|
 |
 |
| 3 Reasons Why Getting an MBA Makes Sense from a Career Standpoint |
 |
| |
|
|
|
|
|
|
If you're looking to further your career in your chosen field or even want to make a change, then getting an MBA makes perfect sense. Not only will an MBA help you to achieve a promotion in your current company, but it will also open the doors to additional job opportunities and a substantial increase in salary. Here are three reasons why studying for an MBA makes sense from a career standpoint.
1. Greater Employment Opportunity & Salary
Employers are looking for new staff who not only have experience in their chosen field, but who also have the skills and knowledge to add value to the company. In fact, in 2016, 96% of employers said that they would prefer to employ business school graduates because of the benefit these people will provide for the company they work for.
Having an MBA sets you apart from other job candidates and gives you a distinct advantage in landing that job. And if you're looking for a promotion in your current place of employment, an MBA shows your employer that you are serious in wanting to further your career with your current company.
Some of the top careers for MBA grads include
• Chief Technology Officer
• Computer and Information Systems Manager
• High-End Management Consultant
• Information Technology Director
• Investment Fund Manager
• Financial Manager
• Marketing Manager
As you can see, these are all managerial type positions and therefore they command an excellent salary. Having an MBA also grants you much more job security, or at least the security of knowing that you have a qualification that is desired by most employers.
2. Specialization in Your Chosen Field
Most MBA programs offer different specializations in the field that you're choosing to work in. Choosing a certain specialization enables you to build a stronger skill base, which makes you extremely attractive to potential employers.
Listing a specialization on your resume helps you to stand out from the crowd of other candidates and therefore employers will view your application more favorably, especially if they need additional skills in that particular field.
3. Attaining Transferable Skills
Not only will an MBA help you to further your prospects in your current company, but if you're thinking of moving into another field entirely, your MBA gives you the opportunity to explore a myriad of other career options.
The skills that you've acquired through your studies, including leadership, analysis, intellectual creativity, communication and critical thinking, can all be applied to many varied careers with an MBA.
So if you're currently working in finance, for example, and down the track you decide that you really want to move into the hospitality industry, your MBA will give you the opportunity to change career paths much more seamlessly.
Getting an MBA these days does not need to be difficult. You don't need to throw in your current job and head back to university full-time and therefore sacrifice your current lifestyle. There are now many opportunities to study for your MBA online while you're still working and looking after your family.
|
|
|
| |
|
|
| |
|
|
|
|
|
|
|
|
|
 |
 |
| Asian Healthcare Market Expected to Overtake Europe |
 |
| |
|
|
| Jan 04, 2017 |
|
Category:
|
|
Tags:
|
|
|
|
|
|
By 2025, regional dynamics will end up altering global healthcare in a variety of ways. Take North America, which is currently the biggest healthcare market in the world, and Europe, which is the second biggest healthcare market. These two markets are expected to change, with one market in another part of the world growing stronger. Keep reading to find out more.
A Growing Asian Healthcare Market
In particular, Asia, not including Japan, has a healthcare market that’s expected to continue expanding at around 11.7%. As a result, it will end up overtaking Europe over time. This opens up even more opportunities for individuals who are interested in studying everything from healthcare administration and healthcare leadership, to healthcare management and everything in between.
Contrast that with Japan, which has the fourth biggest healthcare market. It will be eventually be overtaken by Latin America, which is expected to keep expanding at around 10.4%.
When 2025 rolls around, the regional shares throughout the healthcare market, from the smallest to the biggest, will shift, and the order will end up being: North America followed by Asia and Europe. Those will be followed by Latin America and Japan as the biggest markets.
What’s Driving Growth in the Asian Healthcare Market?
The incredible growth that has been occurring in the healthcare market throughout Asia is actually driven by China and India. There are also funding schemes from governments, and those are expected to add the Philippines, Vietnam, and Indonesia to the list of countries whose healthcare industry is rapidly expanding.
What about Europe?
Europe will fall from its current ranking in the healthcare market. The countries that have been the most adversely affected by a Euro in decline are Spain, Portugal, and Greece. In the meantime, the United Kingdom is trying to deal with the pound’s decline and all of the problems that have arisen since Brexit. And because of political tensions, Russia isn’t doing as well, either.
What Does This Mean for Investors?
As the global healthcare industry begins to shift, those who are stakeholders in this market will have to alter the focus of their investments. Shifting your spotlight to Asia will be necessary, as it really does have the potential to eventually be the biggest healthcare market on the planet, especially if its existing growth rate continues.
Another smart investment opportunity is in the Latin American market, where experts are predicting some important opportunities that are the result of increased demand and a rising population, along with a growing middle class that has more disposable income than before. In that part of the world, growth has also been supported by a boost in private healthcare options, as well as a bigger population that is working and able to support seniors.
Ultimately, it will be wise healthcare investors who will help the Latin American and Asian markets continue expanding. Although there will be a few difficulties along the way, healthcare players can invest in these parts of the world, as well as locate partners and design strategies for each nation, to inspire more growth.
|
|
|
| |
|
|
| |
|
|
|
|
|
|
|
|
|
 |
 |
| The Benefits of Volunteering Abroad for Student Nurses |
 |
| |
|
|
|
|
|
|
Volunteering abroad can be an excellent way for people in all professions to add some experience to their resume and gain some vital insights into what working life could be like for them. If you are hoping to pursue a career in nursing, volunteering in healthcare abroad can be an excellent way for you to gain vital on-the-job experience, get a feel of what working in nursing is like and even devote your time to helping a variety of great causes. Whether you work in a hospital abroad or volunteer your time and skills to helping people who are victims of disasters in other countries, for example, there are many great reasons for nursing students to consider overseas voluntary work as a method of gaining experience.
Work Whilst You Study
Thanks to online and distance learning degree courses which are offered by a range of universities, colleges and nursing schools, it’s never been easier for student nurses to undertake voluntary work abroad whilst they study. For example, you can study for a masters in nursing online from wherever in the world you may be providing that you have a laptop and a working Wi-Fi connection. Because of this, you’re not just limited to overseas voluntary positions which fit around your course; you can tailor your course to fit around them instead.
Gaining Experience
There are many different opportunities for nurses to volunteer abroad, and doing so as a student nurse can help you to gain valuable experience which will look great on your resume and help you out when it comes to landing the graduate nursing job of your dreams. Even though qualified nurses are currently in higher demand than ever before, it’s still important for recently graduated nurses to have plenty of related work experience.
Learn a New Language
For student nurses, volunteering abroad isn’t just about gaining vital medical and healthcare work experience. Volunteering abroad also provides student nurses with a range of other benefits, such as the opportunity to learn a new language – something that can be a hugely beneficial skill in a wide range of different fields. For student nurses, learning another language can be an excellent way to open up further future doors of opportunity when it comes to finding graduate jobs or career progression. Along with a qualification such as RN to MSN programs, having the ability to speak another language can definitely further your prospects.
Networking
When volunteering abroad in the medical and healthcare sector, there will be plenty of opportunities for student nurses to network with professionals in the field and get to know people who can significantly help them when it comes to successfully graduating and going on to pursue a nursing career. When volunteering abroad, student nurses will be working with a range of health professionals, with doctors, nurses, fellow students and more on hand for asking questions and learning new things.
If you’re a student nurse, volunteering abroad can be a great way to get hands-on medical experience and travel at the same time. Volunteering abroad can be hugely beneficial for student nurses, as not only do you gain great experience, you can also learn a new language and meet some awesome people, too.
Photo credit / College of Nursing, The University of Arizona
|
|
|
| |
|
|
| |
|
|
|
|
|
|
|
|
|
 |
 |
| A Quick Look at Healthcare Needs in Asia - Hospitals in Greatest Need |
 |
| |
|
|
| Sep 14, 2016 |
|
Category:
|
|
Tags:
|
|
|
|
|
|
Throughout Asia, there are more overpopulated countries than on any other continent and although the world knows that China has the largest population, it might surprise you to know that within the next couple of decades India is projected to bypass the population of China, becoming the largest country on earth. However, those honors are dubious because at the same time as they are the largest countries on earth they also have the greatest medical needs among which are hospitals.
Welcoming Medical Professionals from the West
Many of these countries in Asia are considered to be developing nations while others are somewhere in between, being categorized as Newly Industrialized Countries which means they are somewhere in their technological development between developing nations and developed nations. These are the countries, these NICs which welcome medical professionals from the West to both teach as would someone with a doctorate in nursing practice and to help staff clinics and hospitals that are in grave need of human and technological resources. Anyone studying for an advanced degree in nursing from online programs like those offered at Bradley University would do well to specialize in something that can be shared with developing and newly industrialized nations in Asia.
Why Hospitals Are in Such Demand
The most important factor to affect healthcare in Asia is the fact that populations are growing faster than communities and the government can build and supply hospitals to meet greater demands. Also, much of the population in parts of the region has not been to a doctor in recent years, sometimes ever in their lives. As a result, care being given would be both acute and chronic. With a lack of healthcare facilities in smaller outlying regions, communicable diseases run rampant and are difficult to stop.
Technology Is Lagging
Since most countries in Asia other than Korea and Japan are really lagging when it comes to technology, the very technology we depend on here in the West is sadly missing from their healthcare systems. This is something governments are focusing on and within the next few years this issue should have less of an impact. From current diagnostics to various technologies used in the treatment of diseases such as cancer, there is a real need for better technology and professionals to operate each piece of equipment. Growing numbers of experts are finding work in Asia and together with a people who are eager to learn, healthcare will get the boost it so sorely needs.
While the population of Asia exceeds that of any other region in the world, the region is still behind in terms of healthcare professionals and healthcare technology. Anyone seeking to advance their careers in medicine would do well to study for an advanced degree and then seek employment in the region. This would provide a wonderful opportunity for personal growth, helping NICs and developing countries expand their technology to serve an already large and continually growing population. Shared resources offer the best chance of success.
|
|
|
| |
|
|
| |
|
|
|
|
|
|
|
|
|
 |
|
|
